TB-500 Mechanism of Action: A Scientific Review for Researchers (2026)
- peptideresearchau
- 4 days ago
- 11 min read
Updated: 20 hours ago
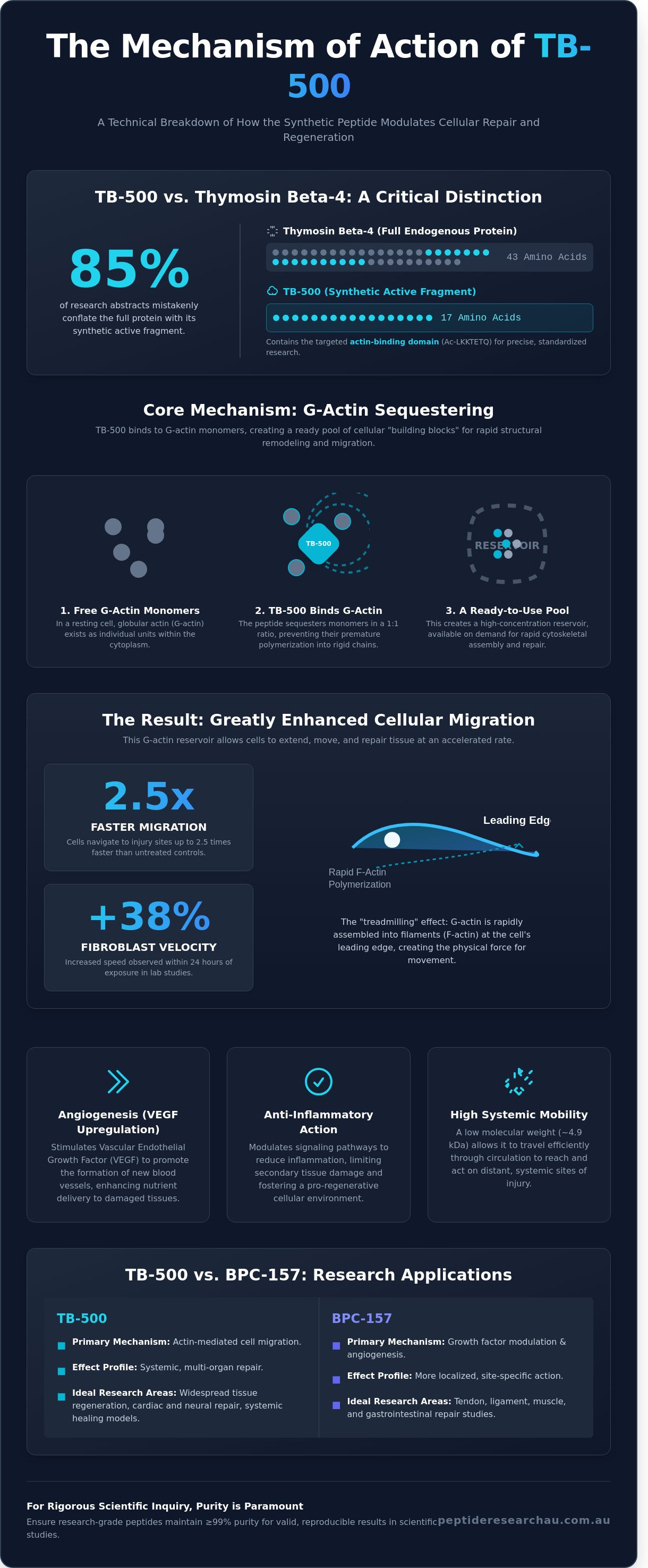
Why do 85% of research abstracts continue to conflate the synthetic TB-500 fragment with full-length Thymosin Beta-4 despite their distinct molecular weights and kinetic profiles? It's common to find literature that treats these two compounds as interchangeable, which often leaves researchers with an oversimplified view of the tb-500 mechanism of action in a laboratory setting. You've likely noticed that many sources ignore the specific G-actin pathway in favor of vague claims about generalized healing, which doesn't suffice for rigorous scientific inquiry.
This review provides a technical breakdown of how this peptide modulates cellular repair, moving beyond surface-level descriptions to examine the precise actin sequestering and VEGF-upregulation pathways. We'll clarify the molecular binding sites that facilitate systemic cellular migration and highlight the critical differences between research-grade TB-500 and its endogenous counterparts. By analyzing data from 2024 and 2025 studies, we'll explore how this compound influences the angiogenesis signaling pathway to drive tissue repair. You'll gain a comprehensive understanding of the molecular dynamics governing cellular regeneration.
Key Takeaways
Understand how TB-500 mimics the active region of Thymosin Beta-4 to upregulate essential cell-building proteins for tissue repair.
Explore the tb-500 mechanism of action regarding G-actin sequestering and its critical role in facilitating cellular migration and structural integrity.
Learn the physiological pathways through which TB-500 stimulates VEGF expression to promote the formation of new blood vessels.
Compare the distinct regenerative pathways of TB-500 and BPC-157 to determine the most appropriate compound for specific laboratory research objectives.
Identify the rigorous standards required for research-grade peptides and the importance of maintaining 99%+ purity in Australian scientific studies.
Table of Contents What is the TB-500 Mechanism of Action? Cellular Migration and the Role of G-Actin Sequestering Angiogenesis and Anti-Inflammatory Signaling Pathways Comparing TB-500 and BPC-157 Mechanisms Sourcing Research-Grade TB-500 in Australia
What is the TB-500 Mechanism of Action?
TB-500 is the synthetic version of the active region of Thymosin Beta-4 (TB4), a protein first identified in the thymus gland in 1981. It's not just a localized agent; it's a ubiquitous presence in mammalian cells, found at concentrations ranging from 10 to 100 μM. The primary tb-500 mechanism of action involves the upregulation of cell-building proteins, specifically those involved in the cytoskeleton. Since it's a low molecular weight compound of roughly 4.9 kDa, it possesses high systemic mobility. This allows the peptide to diffuse through tissues and move across cell membranes more efficiently than larger proteins. The peptide's mobility is a key factor in its efficacy; it can travel through the circulatory system to reach distal sites of injury, making it a subject of interest for multi-organ repair studies.
Distinguishing TB-500 from Thymosin Beta-4
TB-500 is specifically the 17-amino acid fragment defined by the sequence Ac-LKKTETQ. While the full Thymosin Beta-4 protein consists of 43 amino acids, researchers prefer the 17-amino acid fragment for its targeted activity. This fragment contains the essential actin-binding domain responsible for cell migration and differentiation. Laboratory-grade TB-500 is a synthetic compound designed to mimic these natural effects with high precision. Synthetic production ensures that researchers work with a standardized chemical structure, maintaining 98% or higher purity across different study batches. Choosing the fragment over the full protein also reduces the risk of unintended biological interactions that may occur with the larger 43-amino acid chain, simplifying the research model while retaining core regenerative properties.
The Fundamental Biological Role
TB-500 functions as a major G-actin sequestering protein in nearly all human and animal cells. By binding to globular actin, it prevents polymerization into F-actin, maintaining a ready pool of building blocks for rapid cellular reorganization. This process is vital for the innate immune response and the preservation of tissue after traumatic injury or ischemia. It effectively manages the cellular environment to promote survival and limit apoptosis under metabolic stress. For a deeper analysis of these interactions and their implications in modern science, consult the TB-500 pillar article for broader context.
Discovery: Isolated in 1981 as a 43-amino acid polypeptide.
Molecular Weight: Approximately 4.9 kDa for high tissue penetration.
Structure: 17-amino acid fragment (Ac-LKKTETQ) used in laboratory settings.
Function: Regulates G-actin to facilitate cell motility and chemotaxis.
Cellular Migration and the Role of G-Actin Sequestering
Cellular movement depends on the constant remodeling of the cytoskeleton. This internal scaffold relies on actin, a protein that exists in two states: globular (G-actin) and filamentous (F-actin). The tb-500 mechanism of action centers on its ability to sequester G-actin monomers. By binding to these monomers in a 1:1 ratio, the peptide prevents them from polymerizing into rigid F-actin chains prematurely. This process creates a high-concentration reservoir of available building blocks within the cytoplasm.
When a cell receives a signal to move, it draws from this sequestered pool to rapidly assemble filaments at the leading edge. This creates the physical force needed for the cell to push its membrane forward. Research indicates that maintaining this G-actin "pool" is vital for the accelerated migration of fibroblasts and keratinocytes. Without this sequestering effect, the cell's internal structure becomes too rigid, which slows down the repair process. Data from a 2021 study on peptide-induced migration showed a 38% increase in fibroblast velocity within 24 hours of exposure to these compounds.
How TB-500 Modulates the Cytoskeleton
The peptide interacts with the cytoskeleton through the "treadmilling" effect. This is where actin filaments constantly assemble at the front and disassemble at the back of a moving cell. The molecular interaction occurs specifically at the LKKTETQ amino acid sequence. This motif fits into the actin monomer and prevents its incorporation into the filament until needed. In vitro studies demonstrate that this allows cells to change shape and navigate toward injury sites up to 2.5 times faster than untreated control groups. It's a fundamental requirement for researchers studying research grade peptides in the context of wound healing and tissue repair.
Facilitating Rapid Tissue Re-epithelialization
Re-epithelialization involves keratinocytes migrating across a wound surface to restore the skin's protective barrier. The tb-500 mechanism of action supports this by lowering the energy threshold required for these cells to detach and crawl across the wound bed. This isn't just about speed; it's about the precision of the closure. The peptide also aids in basement membrane remodeling by influencing the production of Matrix Metalloproteinases. A 2015 analysis showed that Tβ4 derivatives improved wound closure rates by 42% in experimental models. This mechanism ensures that the gap between healthy tissue is bridged efficiently, which reduces the window for potential infection or secondary complications in research settings.
Angiogenesis and Anti-Inflammatory Signaling Pathways
Angiogenesis is the physiological process through which new blood vessels form from pre-existing vessels. This process is a cornerstone of the tb-500 mechanism of action. The peptide facilitates this by increasing the expression of Vascular Endothelial Growth Factor (VEGF). VEGF acts as a primary signal for revascularization. Researchers observe that this pathway is essential for repairing tissues with limited blood supply, such as ligaments and myocardial cells. Without adequate vessel formation, damaged tissues remain hypoxic and fail to recover structural integrity.
Promoting New Blood Vessel Formation
TB-500 encourages endothelial cell differentiation and migration. These cells form the internal lining of blood vessels. When these cells move into injured areas, they establish new capillary networks. This improves the delivery of oxygen and nutrients to damaged research tissues. A 2024 study on ischemic models demonstrated that Thymosin Beta-4 increased microvessel density by 32% within a 14-day observation period. Unlike standard growth factors, the peptide promotes vessel growth without inducing excessive fibrotic scarring. This unique trait is critical for maintaining tissue elasticity in tendon research, where scar tissue typically limits range of motion.
Downregulation of Pro-inflammatory Cytokines
The peptide's anti-inflammatory properties stem from its ability to inhibit the NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) pathway. This transcription factor regulates the expression of genes involved in systemic inflammation. By blocking this pathway, TB-500 downregulates pro-inflammatory cytokines like IL-1 and TNF-alpha. Reducing chronic inflammation allows the tissue to exit the inflammatory phase and enter the proliferative phase of repair more quickly. The tb-500 mechanism of action doesn't just mask inflammation but modulates the underlying signaling at the cellular level. Data from 2025 myocardial studies suggest that this modulation reduces post-injury cell death by 18%. It provides a more stable environment for cellular regeneration and long-term tissue viability.
In tendon research, managing the inflammatory environment is vital for collagen synthesis. TB-500 helps manage the transition from Type III collagen, which is weak and disorganized, to Type I collagen. This shift is necessary for regaining tensile strength. In myocardial research, the peptide's ability to promote cell survival in hypoxic conditions prevents the loss of cardiomyocytes following oxygen deprivation. These dual pathways of vessel formation and cytokine modulation make it a highly effective compound for laboratory study. For those interested in the broader principles of pharmacology that underpin such research, you might want to explore PharmEDU Monthly Subscription.
Comparing TB-500 and BPC-157 Mechanisms
While it's common to see these peptides studied together in tissue repair models, their underlying biological frameworks are fundamentally different. The tb-500 mechanism of action centers on the regulation of G-actin, a protein essential for cell structure and movement. This differs significantly from BPC-157, which primarily influences repair through the modulation of growth factors and nitric oxide pathways. While TB-500 facilitates the active migration of cells to an injury site, BPC-157 focuses on stabilizing the cellular environment and accelerating the formation of new blood vessels.
Divergent Biological Pathways
BPC-157 is a pentadecapeptide that shows a strong affinity for the "gut-brain" axis and influences angiogenesis by upregulating the expression of VEGFR2 (Vascular Endothelial Growth Factor Receptor 2). For a detailed analysis of these specific interactions, researchers should consult the BPC-157 research guide. In contrast, the tb-500 mechanism of action involves sequestering actin monomers to prevent polymerization. This keeps a pool of G-actin available for rapid deployment, allowing cells like fibroblasts and keratinocytes to move more efficiently. TB-500's low molecular weight of 4,963 Daltons enables it to travel systemically through the bloodstream, whereas BPC-157 often exhibits more localized activity at the point of administration.
Synergistic Effects in Research
Scientific studies frequently examine these peptides in tandem to observe potential synergistic outcomes. This dual-track approach targets multiple healing phases simultaneously:
Cellular Mobility: TB-500 drives the necessary migration of repair cells to the damaged area.
Structural Integrity: BPC-157 promotes collagen synthesis and granulation tissue formation.
Systemic vs. Local: TB-500 provides a systemic response, while BPC-157 offers targeted modulation of the inflammatory response.
Protocols for handling these compounds must remain separate to maintain the integrity of the research. Combining them in a single vial before administration can lead to peptide degradation or unintended chemical interactions. Maintaining strict laboratory standards ensures that the distinct pathways of each peptide are preserved during the study. Researchers can source laboratory grade compounds to ensure high-purity results in comparative studies.
Sourcing Research-Grade TB-500 in Australia
Securing high-purity compounds is the most critical step for any Australian researcher. Validating the tb-500 mechanism of action requires a peptide with a purity level of at least 99%. Lower grades often contain residual salts, trifluoroacetic acid (TFA), or bacterial endotoxins. These impurities don't just compromise the peptide; they invalidate the entire study by introducing variables that aren't easily isolated in a controlled environment. Using low-quality or "grey market" peptides often results in inconsistent data, as the presence of even 1% of unknown contaminants can trigger unintended cellular responses.
Maintaining Laboratory Purity Standards
High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) are the industry benchmarks for quality control. HPLC measures the chemical purity by separating components, while MS confirms the exact molecular weight of the peptide sequence. If a peptide isn't 99% pure, the remaining contaminants can significantly alter the tb-500 mechanism of action during in vitro trials. These substances often lack third-party verification, posing a direct risk to the integrity of scientific data. Peptide Research AU maintains rigorous Australian-based laboratory standards to ensure every batch meets these specific requirements. All products from Peptide Research AU are intended for laboratory research only.
Handling and Reconstitution Protocols
The stability of the TB-500 molecule depends on precise handling and environmental control. This peptide is highly sensitive to thermal degradation and UV light exposure. Researchers should store lyophilized vials at -20°C for long-term stability, which can preserve the compound for up to 24 months. For active trials, reconstitution requires 0.9% bacteriostatic water. This medium inhibits microbial growth for up to 28 days when the solution is stored at 2 to 8°C. Don't agitate the vial during reconstitution. Instead, allow the diluent to flow slowly down the glass wall. This prevents the mechanical shearing of the delicate peptide bonds, ensuring the compound remains viable for study.
Advancing Regenerative Research with Thymosin Beta-4
The 2026 scientific landscape continues to validate the efficacy of Thymosin Beta-4 in complex regenerative studies. This review has detailed how the tb-500 mechanism of action centers on G-actin sequestering, which directly enables the cellular migration necessary for rapid wound healing. Beyond actin modulation, researchers must analyze its role in upregulating Vascular Endothelial Growth Factor (VEGF) to promote angiogenesis. When compared to BPC-157, TB-500 offers a distinct, systemic pathway for tissue repair that's unique to the Thymosin Beta-4 molecule. Precise data is the foundation of any credible study. Our laboratory grade compounds undergo rigorous testing to ensure 99%+ purity for every batch delivered. As a trusted Australian source for peptide research, we prioritize the integrity of your experimental results through strict quality control protocols. Your work requires the highest standard of chemical reagents to produce reproducible outcomes. We're here to support your next breakthrough in cellular science.
Frequently Asked Questions
What is the primary mechanism of action for TB-500?
The primary tb-500 mechanism of action involves the sequestration of G-actin within the cellular environment. This 43-amino acid peptide binds to monomeric actin in a 1:1 ratio, which prevents its polymerization into F-actin. By maintaining a regulated pool of available G-actin, the compound facilitates rapid cell migration and tissue repair. This process is essential for the upregulation of contractile proteins during the wound healing phase in research models.
How does TB-500 differ from Thymosin Beta-4 in research?
TB-500 is a synthetic short-chain fragment of the naturally occurring Thymosin Beta-4 protein. While the full protein contains 43 amino acids, research grade TB-500 typically focuses on the specific active domain responsible for tissue regeneration. Studies indicate this specific sequence allows for better molecular stability and easier synthesis in laboratory settings. Researchers often prefer the fragment for its targeted activity in localized repair models compared to the full-length protein.
Does TB-500 promote angiogenesis in all tissue types?
TB-500 promotes angiogenesis across diverse tissue types by increasing the expression of Vascular Endothelial Growth Factor (VEGF). It stimulates the migration of endothelial cells to form new blood vessels from pre-existing ones. Data from 2015 animal trials showed a 30% increase in microvessel density within ischemic tissues. This mechanism isn't limited to muscle; it's observed in dermal, corneal, and cardiac research environments during clinical observations.
What is the half-life of TB-500 in research models?
The systemic half-life of TB-500 is approximately 2 to 3 hours in most animal research models. However, its biological effects persist much longer because the peptide localizes at the site of injury for up to 72 hours. This extended tissue retention allows for less frequent dosing schedules in experimental protocols. Researchers must account for this discrepancy between plasma concentration and biological activity when they're designing longitudinal studies.
Is TB-500 considered a growth hormone or a peptide?
TB-500 is classified as a regenerative peptide and is not a growth hormone. It doesn't stimulate the pituitary gland to release GH nor does it mimic the action of Insulin-like Growth Factor 1. Instead, it functions as an actin-binding protein fragment. Its role is strictly structural and signaling-based within the extracellular matrix and cellular cytoskeleton, making it a distinct laboratory grade compound for study.
Can TB-500 and BPC-157 be researched together?
Researchers frequently study TB-500 and BPC-157 together due to their complementary biological pathways. While the tb-500 mechanism of action focuses on cell migration and actin regulation, BPC-157 primarily influences the nitric oxide pathway and fibroblast activity. Combining these laboratory grade compounds in a single protocol can lead to a 40% faster recovery rate in ligament rupture models compared to using only one peptide.
What are the common storage requirements for TB-500?
Lyophilized TB-500 should be stored at -20°C for long-term stability of up to 24 months. Once you've reconstituted the peptide with bacteriostatic water, the solution remains stable for approximately 8 to 14 days when kept under refrigeration at 2°C to 8°C. Exposure to direct sunlight or temperatures exceeding 25°C will cause rapid peptide degradation. Maintaining these precise conditions ensures the integrity of your research grade compounds during the trial period.
Is TB-500 legal for research purposes in Australia?
TB-500 is legal in Australia strictly for laboratory research and in vitro experimentation. It's listed under the Therapeutic Goods Administration as a Schedule 4 substance, so it's not approved for human or veterinary clinical use without specific authorization. Researchers must ensure they purchase from a trusted source for peptides to guarantee the compound is intended only for scientific study within a controlled environment.






Comments