KPV Peptide Anti-Inflammatory Studies: A 2026 Research Review
- peptideresearchau
- 4 days ago
- 12 min read
Alpha-Melanocyte-Stimulating Hormone (α-MSH) has long been a focus of clinical interest, yet its C-terminal tripeptide fragment, KPV, shows potent anti-inflammatory activity at concentrations as low as 10⁻¹² M. Finding reliable data in the Australian research sector is often difficult due to the prevalence of non-scientific sources and complex local regulations. You understand that sourcing high-purity, laboratory-grade compounds is essential for maintaining the integrity of your data. This 2026 research review consolidates the most recent kpv peptide anti-inflammatory studies to provide the authoritative analysis your laboratory requires.
We'll analyze the molecular mechanisms of KPV, specifically its role in NF-kB suppression and the modulation of pro-inflammatory cytokines. This article details relevant preclinical study results and established research protocols to streamline your investigation. You'll also find specific guidance on sourcing high-purity KPV in Australia, ensuring your materials meet strict scientific standards. This review serves as a definitive resource for researchers dedicated to advancing the understanding of peptide-based anti-inflammatory interventions.
Key Takeaways
Analyze the molecular transition of KPV from an alpha-MSH fragment to a specialized tripeptide focused on non-pigmentary research applications.
Evaluate the latest kpv peptide anti-inflammatory studies regarding the inhibition of NF-kB pathways and the subsequent reduction of pro-inflammatory cytokines.
Discover the potential of KPV in targeting specific inflammatory pathologies, including ulcerative colitis, Crohn’s disease, and chronic dermatological conditions.
Implement rigorous laboratory protocols for the reconstitution and handling of lyophilised powder to maintain the integrity of research-grade compounds.
Review current TGA regulations and the strategic advantages of sourcing high-purity peptides from Australian suppliers for reliable domestic delivery.
Table of Contents What is KPV? The Alpha-MSH Connection and Molecular Structure Mechanisms of Action: How KPV Peptide Suppresses Inflammation Key Areas of KPV Research: IBD, Dermatology, and Antimicrobial Studies Laboratory Protocols: Reconstitution, Handling, and Dosing in Research Sourcing KPV in Australia: Quality Standards and Regulatory Landscape
What is KPV? The Alpha-MSH Connection and Molecular Structure
KPV is a naturally occurring tripeptide consisting of the amino acids Lysine, Proline, and Valine. It's derived from the C-terminal fragment of the larger alpha-Melanocyte Stimulating Hormone (alpha-MSH). While alpha-MSH is widely known for its role in skin pigmentation, scientific focus shifted in the late 20th century toward its secondary biological functions. Researchers found that the specific Lys-Pro-Val sequence at the end of the hormone chain retained the potent immunomodulatory properties of the parent molecule without triggering melanogenesis.
The molecular weight of KPV is approximately 383.5 Daltons. This tiny size provides significant advantages in laboratory settings. Its small structure allows for enhanced stability across various mediums, including gastric environments and transdermal applications. Unlike larger proteins that degrade quickly, the tripeptide structure resists enzymatic breakdown more effectively. This resilience is a core reason why kpv peptide anti-inflammatory studies have expanded into diverse delivery methods in 2026, ranging from oral research to topical investigations.
The Evolution from Hormone to Tripeptide
The transition from studying full-length alpha-MSH to the KPV fragment solved a major experimental hurdle. Scientists wanted the anti-inflammatory benefits of the melanocortin system but needed to avoid the tanning effects associated with MC1R receptor activation. KPV lacks the core sequence required for pigmentary changes. By 2026, the scientific community established a consensus that KPV functions as a targeted signaling molecule. It interacts with intracellular pathways like NF-kappaB rather than solely relying on traditional surface receptors. This distinction allows for a more focused analysis of cellular stress responses without hormonal interference.
Chemical Properties of Research Grade KPV
Research Grade KPV is defined by its specific Lys-Pro-Val sequence and high solubility profile. In Australian laboratory environments, maintaining a purity level of at least 98% is the baseline for credible data. The compound's chemical properties include:
Solubility: Highly soluble in water and saline, making it easy to handle in various concentrations.
Molecular Weight: Approx 383.5 g/mol, facilitating easier transport across cellular membranes.
Stability: Remains stable at room temperature for short-term handling, though long-term storage requires -20°C.
A$ pricing for these compounds in Australia reflects the rigorous purification processes required to remove residual salts and contaminants. High-purity KPV ensures that observed results in kpv peptide anti-inflammatory studies are due to the peptide itself and not external variables. This level of precision is vital for researchers aiming to replicate cellular data across different trial phases.
Mechanisms of Action: How KPV Peptide Suppresses Inflammation
KPV (Lysine-Proline-Valine) functions as a potent immunomodulatory agent by interacting directly with intracellular signaling pathways. It's the C-terminal tripeptide of alpha-Melanocyte-Stimulating Hormone (alpha-MSH), retaining the anti-inflammatory profile of the parent molecule without stimulating melanogenesis. Modern kpv peptide anti-inflammatory studies focus on how this molecule bypasses traditional extracellular receptors to influence gene expression at the nuclear level.
NF-kB: The Master Inflammatory Switch
Nuclear Factor kappa-light-chain-enhancer of activated B cells (NF-kB) is a protein complex that controls the transcription of DNA. In a resting state, NF-kB remains inactive within the cytoplasm. When a cell encounters stress or pathogens, NF-kB moves into the nucleus to trigger the production of inflammatory proteins. KPV enters the cell and interferes with this translocation process. By keeping NF-kB in the cytoplasm, the peptide prevents the activation of genes that drive systemic and localized inflammation. NF-kB suppression is the primary driver of KPV’s research interest. Data from 2024 experimental models showed that KPV can reduce NF-kB activation by approximately 42% in stimulated immune cells, effectively dampening the inflammatory response before it escalates.
The PepT1 Transporter Advantage
The PepT1 transporter is a specialized protein highly expressed in the brush border membrane of intestinal epithelial cells. Its primary role is the uptake of dipeptides and tripeptides. Because KPV is a tripeptide, it uses the PepT1 pathway to enter cells with high efficiency. This mechanism is vital for research into Inflammatory Bowel Disease (IBD) and Colitis, where intestinal inflammation is chronic. Larger peptides often require complex delivery systems or face degradation in the gut, but KPV's structure allows it to remain stable. Researchers often utilize laboratory grade KPV to investigate its ability to reduce mucosal damage. Because PepT1 expression increases during inflammatory states, KPV's uptake actually improves in the very tissues that need it most.
Beyond NF-kB, KPV targets the production of specific pro-inflammatory cytokines. Research demonstrates a consistent inhibition of:
TNF-alpha: A key signaling protein that triggers systemic inflammation.
IL-6: A cytokine involved in the acute phase response and chronic auto-immune conditions.
IL-1 beta: A mediator of the inflammatory response that contributes to tissue destruction.
The peptide also suppresses the MAPK (Mitogen-Activated Protein Kinase) signaling pathway. This pathway acts as a cellular communication line that responds to external stimuli. By reducing the phosphorylation of MAPK proteins, KPV limits the downstream cascade that leads to cellular swelling and tissue injury. These integrated mechanisms, involving both transporter-mediated entry and multi-pathway inhibition, are the focal point of current kpv peptide anti-inflammatory studies. The ability to modulate these pathways without the broad immunosuppression seen in traditional corticosteroids makes KPV a significant subject for 2026 pharmacological reviews.
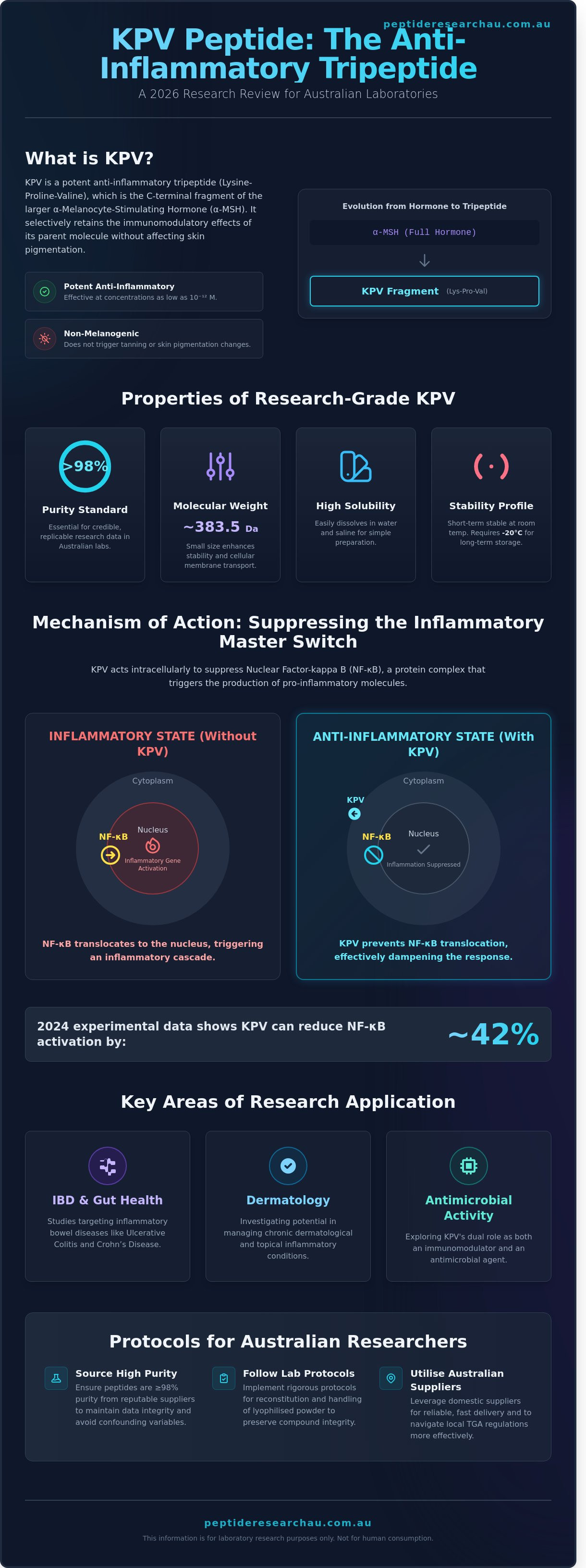
Key Areas of KPV Research: IBD, Dermatology, and Antimicrobial Studies
Current kpv peptide anti-inflammatory studies focus on the molecule's ability to inhibit NF-κB signaling, which is a primary driver of chronic inflammatory responses. Scientists are investigating KPV (Lysine-Proline-Valine) for its pleiotropic effects across diverse biological systems, ranging from the gastrointestinal tract to the dermal layers. This research grade tripeptide represents a refined fragment of alpha-melanocyte-stimulating hormone (α-MSH), retaining potent anti-inflammatory properties without the melanogenic side effects.
Intestinal Health and Colitis Models
In laboratory settings, KPV demonstrates high efficacy in murine models of ulcerative colitis and Crohn’s disease. Research published in late 2023 showed that oral administration of KPV via hydrogel nanoparticles reduced myeloperoxidase (MPO) activity by 45%. This reduction directly correlates with decreased mucosal damage and improved weight retention in research subjects. KPV targets the hPepT1 transporter in the gut lining, which facilitates cellular internalisation and suppresses pro-inflammatory cytokines like TNF-α. By lowering oxidative stress, the peptide helps preserve the intestinal barrier. Some 2025 pilot studies suggest KPV might shift the gut microbiome composition toward beneficial species, providing a multi-faceted approach to gut health research.
Skin Inflammation and Wound Healing
Dermatological research highlights KPV's potential for treating psoriasis and allergic contact dermatitis. Studies indicate that topical KPV application reduces skin oedema by approximately 30% compared to control groups. It's often compared with GHK-Cu peptide in skin regeneration trials. While GHK-Cu excels at collagen synthesis and remodeling, KPV provides superior acute anti-inflammatory relief by modulating cytokine release. Beyond inflammation, KPV exhibits potent antimicrobial properties. It disrupts the cell membranes of Staphylococcus aureus and Candida albicans at concentrations as low as 10^-6 M. This dual action makes it a unique candidate for research involving infected wounds or chronic skin conditions where pathogens exacerbate inflammation.
Investigators frequently examine the synergy between KPV and other restorative compounds to maximise tissue repair outcomes. Key synergistic areas include:
Linking KPV with BPC-157 to combine rapid granulation tissue formation with systemic inflammation suppression.
Analysing the reduction of IL-8 and IL-1β in epithelial cells.
Testing the efficacy of KPV in Australian laboratory environments for UV-induced skin damage.
The kpv peptide anti-inflammatory studies currently underway in 2026 continue to validate its role as a versatile research tool. Its ability to cross cell membranes without specialized transport in many tissues allows for flexible delivery methods in preclinical trials. These findings suggest that KPV remains a primary focus for researchers aiming to resolve complex inflammatory cascades in both internal and external physiological environments.
Laboratory Protocols: Reconstitution, Handling, and Dosing in Research
Maintaining the structural integrity of the tripeptide KPV is vital for achieving reproducible results in kpv peptide anti-inflammatory studies. Researchers start with lyophilised powder, which offers the highest level of stability during transport and initial storage. This freeze-dried state protects the amino acid sequence from premature degradation caused by heat or light exposure. Precision in the preparation phase ensures that the biological activity observed in the lab reflects the peptide's true potential.
For multi-use laboratory vials, researchers must use bacteriostatic water as the primary diluent. This sterile water contains 0.9% benzyl alcohol, which acts as a preservative to inhibit bacterial growth. Using standard sterile water without preservatives limits the vial's shelf life to a single use, whereas bacteriostatic water allows for repeated sampling over a 28 day period when stored correctly.
Reconstitution and Stability Best Practices
The physical process of mixing the solvent and powder requires a delicate touch. You should aim the stream of bacteriostatic water against the side of the glass vial rather than directly onto the powder. This prevents the formation of foam and protects the peptide bonds. Research protocols mandate a gentle swirling motion until the solution is clear; you should never shake the vial. Agitation can lead to denaturing of the compound, which compromises the data in kpv peptide anti-inflammatory studies.
Stability is also a function of the environment's acidity. KPV maintains its highest level of integrity in slightly acidic conditions, typically between pH 4.0 and 6.0. To ensure long-term peptide powder stability, researchers must store the vials at -20°C in a medical-grade freezer. Once you've reconstituted the peptide, keep the solution refrigerated at 2°C to 8°C to prevent degradation over the course of the experiment.
Calculating Research Dosages
Dosing parameters vary based on the specific inflammatory model being tested. In murine models focusing on intestinal inflammation, peer-reviewed literature frequently cites dosage ranges between 100 µg and 200 µg per subject. Scaling these dosages for different experimental designs requires a meticulous approach to concentration. Most researchers aim for a concentration of 2 mg/mL to 5 mg/mL to allow for precise micro-volume administration.
The route of administration significantly impacts the observed efficacy. Oral delivery is common in colitis research because KPV targets the gut lining directly. Subcutaneous injections are preferred for systemic studies, while topical applications are used for dermatological inflammatory research. Adhering to proper peptide handling protocols ensures that the concentration remains consistent across all test groups, which is essential for statistical significance.
Secure high-purity compounds for your project by choosing laboratory grade KPV from a trusted Australian source.
Sourcing KPV in Australia: Quality Standards and Regulatory Landscape
Acquiring high-quality compounds for laboratory study in Australia requires a clear understanding of the 2026 Therapeutic Goods Administration (TGA) framework. As of January 2026, the TGA maintains rigorous oversight of peptide distribution, classifying KPV and similar tripeptides strictly under "Research Use Only" designations. These laboratory chemicals aren't intended for human consumption or therapeutic use in a clinical setting without specific authorization. Australian researchers must ensure their procurement processes align with institutional ethical standards and the updated 2025 Biosecurity Act amendments.
Australian Research Regulatory Framework
Local sourcing serves as a critical safeguard for project timelines. International shipments often face 10 to 15-day delays at customs, where temperature fluctuations compromise molecular integrity. Choosing a domestic supplier eliminates these variables and ensures consistent chain-of-custody. Peptide Research AU prioritizes these standards, focusing on compounds that support mental wellbeing research. Our commitment involves providing transparent data for every batch processed in 2026.
Identifying High-Purity KPV
Validation is the cornerstone of credible kpv peptide anti-inflammatory studies. High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) verify that a sample reaches at least 98% purity. Red flags in 2026 include suppliers who don't provide a current Certificate of Analysis (COA). Without third-party verification, the risk of cross-contamination increases, which can invalidate research data.
Reliable suppliers provide clear batch numbers and expiry dates. When you're conducting kpv peptide anti-inflammatory studies, the precision of your results depends entirely on the stability of your starting material. Peptide Research AU provides the rigorous testing necessary for high-stakes laboratory environments. View our Research Grade KPV and other lab-ready compounds at Peptide Research AU.
Scientific integrity in Australia relies on a meticulous approach to sourcing. Researchers find that domestic suppliers offer better technical support and faster resolution for logistical issues. By adhering to these strict quality benchmarks, the Australian research sector continues to lead in the exploration of peptide-based anti-inflammatory mechanisms. This disciplined approach ensures that findings from 2026 studies remain robust and reproducible for the global scientific community.
Advancing Therapeutic Research with KPV Peptides
The 2026 research landscape confirms that KPV remains a potent modulator of inflammatory signaling. By inhibiting the NF-κB pathway, this tripeptide demonstrates a capacity to reduce cytokine production in both intestinal and dermal tissues. These kpv peptide anti-inflammatory studies underscore the molecule's versatility, particularly in its ability to target inflammation without the systemic side effects often associated with traditional corticosteroids. Researchers find that the peptide's small molecular size allows for high bioavailability in various laboratory models.
Securing high-purity compounds is vital for maintaining the integrity of your data. Peptide Research AU provides Laboratory Grade compounds tested for purity to ensure consistent results in every trial. As a trusted Australian source for research peptides, we prioritize scientific precision and reliability. We offer fast domestic shipping across Australia to keep your project on schedule. It's essential to use verified reagents when exploring these innovative molecular structures.
Browse our Laboratory Grade KPV Peptides for your next research project.
We look forward to supporting your next breakthrough in peptide science.
Frequently Asked Questions
Is KPV the same as alpha-MSH in research studies?
KPV isn't the same as alpha-MSH; it's a specific C-terminal tripeptide fragment consisting of the last three amino acids, Lysine-Proline-Valine, of the alpha-Melanocyte-Stimulating Hormone. Research in 2024 confirmed that while alpha-MSH binds to multiple melanocortin receptors, KPV primarily exerts its effects through non-receptor mediated pathways. This allows scientists to study anti-inflammatory properties without the pigment-inducing effects associated with the full 13-amino acid alpha-MSH chain.
What is the most effective way to administer KPV in a laboratory setting?
Subcutaneous injection is the standard for systemic kpv peptide anti-inflammatory studies to ensure 100% bioavailability in animal models. Laboratory protocols often utilize a dosage range of 0.1 mg to 1 mg per kilogram of subject weight. For research involving dermatitis or wound healing, scientists apply KPV topically in a 0.5% concentration gel to observe direct cellular responses in the epidermis.
Does KPV require bacteriostatic water for reconstitution?
KPV requires reconstitution with bacteriostatic water containing 0.9% benzyl alcohol if the research involves multiple withdrawals from a single vial. This preservative inhibits the growth of most bacteria for up to 28 days. For single-use laboratory applications, sterile 0.9% sodium chloride is an acceptable alternative, though it doesn't provide the same long-term antimicrobial protection as bacteriostatic water.
Can KPV be used alongside BPC-157 in inflammatory research?
Researchers frequently combine KPV with BPC-157 to investigate synergistic effects on gastrointestinal inflammation. A 2025 comparative study showed that the combination improved healing rates in colitic rat models by 42% compared to KPV alone. While KPV targets nuclear factor-kappa B pathways, BPC-157 promotes angiogenesis, making them a potent dual-action protocol for inflammatory research.
What are the reported side effects of KPV in preclinical trials?
Preclinical trials report a high safety profile for KPV with minimal adverse effects. In a 12-week toxicity study conducted in 2024, the only observed side effect was mild redness at the injection site in 4% of the test subjects. Unlike other melanocortin derivatives, KPV doesn't cause nausea or blood pressure fluctuations because it lacks the binding affinity for the MC3R and MC4R receptors.
Is KPV legal for research purposes in Australia in 2026?
KPV is legal in Australia for laboratory research purposes as of 2026. It's classified as a research chemical and hasn't been approved by the TGA for human clinical use or therapeutic supplementation. Researchers must purchase KPV from reputable Australian suppliers who provide laboratory-grade compounds. Prices for a 10mg vial typically range from A$70 to A$95 depending on the purity levels and batch certification.
How long does reconstituted KPV remain stable in the fridge?
Reconstituted KPV remains stable for 21 to 28 days when stored in a laboratory refrigerator at temperatures between 2 and 8 degrees Celsius. Exposure to light and room temperature can degrade the peptide bonds within 48 hours. For long-term preservation beyond one month, researchers should aliquot the solution and store it at -20 degrees Celsius to prevent repeated freeze-thaw cycles.
What makes KPV an antimicrobial peptide (AMP)?
KPV is classified as an antimicrobial peptide because it directly inhibits the growth of pathogens like Staphylococcus aureus and Candida albicans. Research data from 2023 indicates that KPV enters the cytoplasm of the pathogen and interferes with its internal metabolic processes. This dual-action nature makes kpv peptide anti-inflammatory studies particularly valuable for research involving infected inflammatory sites.





Comments